The History of HIV Cure Research: The Latest Research in BioPharma

Contributing Author: Gina Hagler
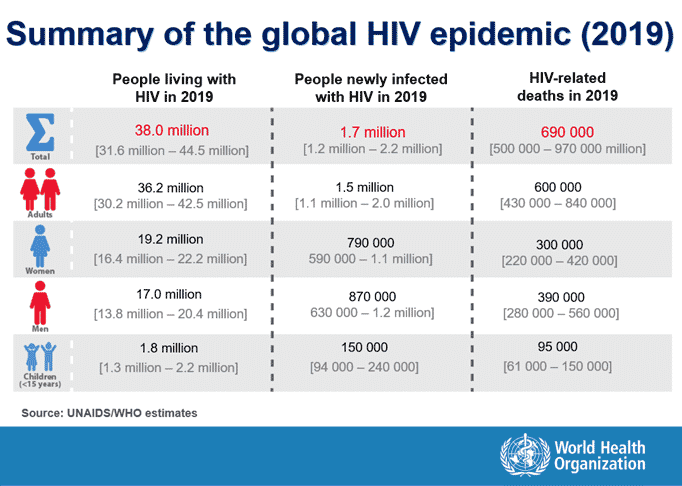
In the forty years since the HIV/AIDS pandemic began in 1981, researchers have learned a lot about this disease that destroys the immune system. Early on, they identified the method of transmission and the means of HIV replication. They went on to successfully turn what had been a deadly disease into a chronic illness through the daily use of antiretroviral drugs for the treatment of HIV. (See The History of HIV Cure Research: Discovery to Treatment to read more.) The use of daily antiretroviral therapy (ART) slowed the rate of new infections since ARTs were made available for use on a global scale. The "Berlin Patient" in 2008 and the "London Patient" in 2019, proved a sterilized cure in which no trace of the virus is left, was possible when they were cured of HIV through the use of stem cell transplants. In 2015, UNAIDS called for the eradication of the virus through the daily use of ARTs to achieve undetectable levels of the virus in 90% of those who are HIV-positive and using HIV medication. Today, the HIV pandemic is still far from over. There is an effective treatment, but still no cure for the 38 million people living with HIV worldwide and those who will be infected in the years to come.
A functional HIV cure has the greatest chance of success
The two patients cured to date were cured with a sterilized cure. A functional cure is another HIV cure strategy. The end result of either is a patient without HIV/AIDS. Yet, the path to the cure is very different:
- Sterilizing Cure: A Treatment where the replication-competent HIV provirus is completely eliminated from the body. With this type of cure, there would be no concern about latency. However, at this time, a sterilized cure is only possible if the HIV-positive person’s immune system is “wiped clean” and restarted with new bone marrow. In the case of the “Berlin” and “London” patients, stem cell transplants were used to cure them of blood cancers. The transplants made use of bone marrow from CCR5--Δ32 donors. These donor cells had a mutation that “renders cells highly resistant to HIV-1 infection,” according to a 2007 peer-reviewed article in BioMed Research International. The transplants cured the patients of both their blood cancers and HIV, proving that a sterilized cure is possible. The article states that “However, it is very difficult to find donors with human leukocyte antigens (HLA) identical to those of recipients for CCR5--Δ32 stem cell transplantation, while the mortality rate of transplant surgery is up to 30%.” The possibility of a host vs. graft reaction, in conjunction with the 30% mortality rate, makes bone marrow transplant surgery a poor route to a cure and it is not being pursued at this time.
- Functional Cure: A treatment where there is durable suppression of viral replication in the absence of antiretroviral therapy. The viral load in a functional cure will permanently remain below the level at which it is possible to either infect others or for the patient to become infectious again from their own viral reservoir. The patient will not progress to AIDs and may be immune to re-contracting the virus from others. The current preference is for a functional cure.
Biopharma holds the promise of a functional cure for HIV through immunology.
Biopharma works with a person’s cells and genes to effect a cure. Rather than make use of toxic drugs with damaging side effects and the necessity of ongoing treatment, gene and cell therapy “updates” the patient’s immune system to fight the virus in the same way that computer software is updated. Jeff Galvin, AGT CEO explains the rationale behind using viruses to fight themselves in this way, “think about this in terms of your computer. We convert those [human] viruses to updates. In other words, we take these things that will invade your body, and reprogram your cells to do all sorts of horrible things - we’re talking about not just colds and cases of flu, but HIV infection. The virus goes into your body, reprograms your cells, and takes over your body over time. But we can go ahead now and split open these deadly viruses, pull out the viral DNA, and put in whatever DNA we want. These are little software updates to the organic computer that is the human cell.” Given biotech’s ability to “reboot” the human operating system through the use of cell and gene therapy, researchers are focused on a functional cure.
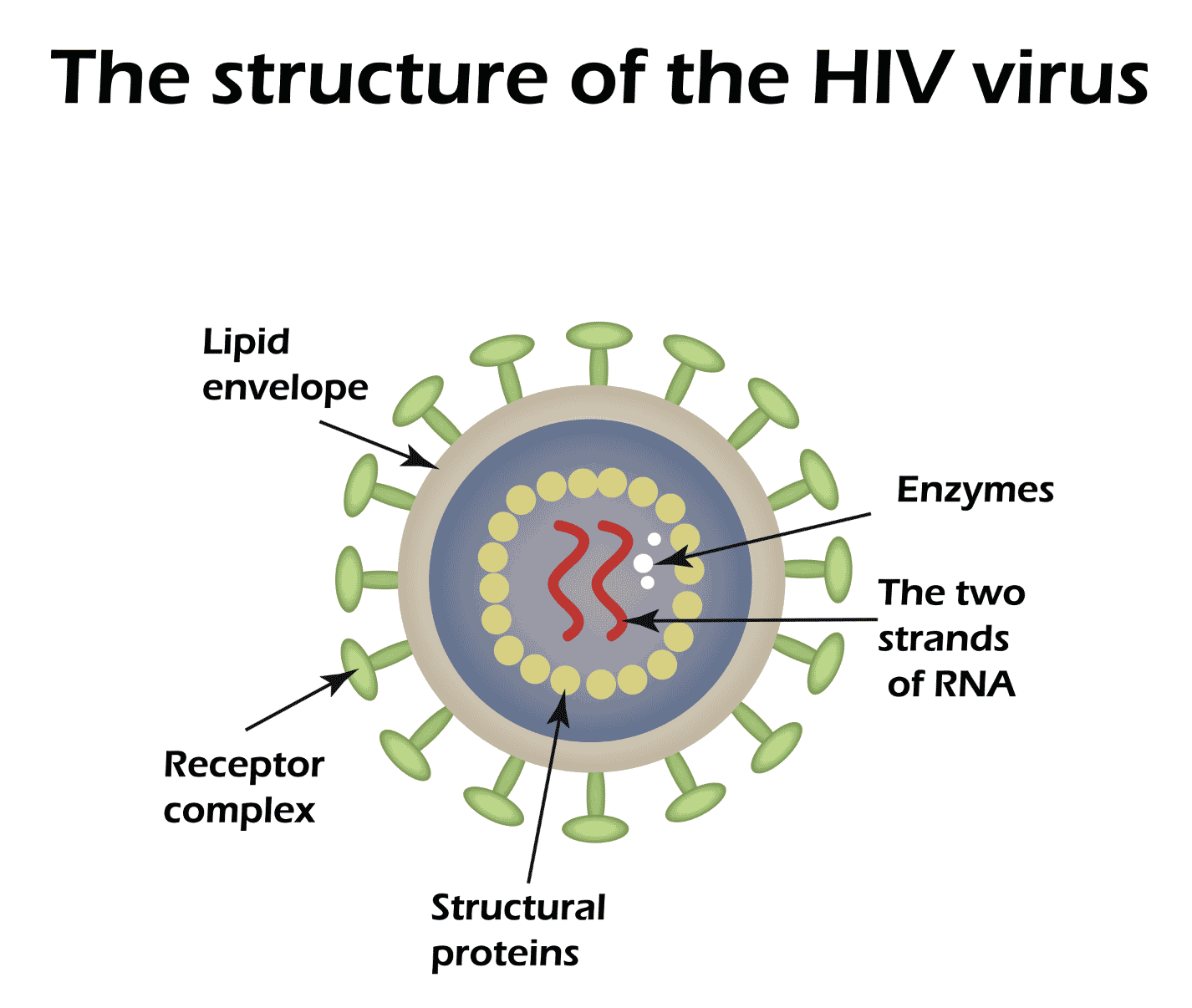
Biopharma is ideally suited to creating a functional cure. Researchers will pull out the HIV DNA from a patient’s cells and use those cells in an autologous cure, i.e., use the patient’s own cells. Once they remove the infected cells, researchers prep them and reintroduce these “updated” cells through infusion. To produce a functional cure for HIV/AIDS, lentiviral vectors can be used in a single-dose. Ideally it will be one-and-done: The patient would be cured, with no additional treatments and no need for the ongoing use of ARTs.
Pauza Presentation
In his August 31st presentation at the Advanced Therapies Congress & Expo 2020, AGT’s Chief Science Officer David Pauza spoke about the latest research in cell and gene therapy for immunotherapy to create a functional cure for HIV. In his remarks, he not only touched on the work being done by AGT on AGT103-T but also noted the way in which AGT examined the work done by others in the field in order to spot strengths and deficiencies in their approaches.
Previous Research
A review of previous HIV gene therapy studies to spot strengths and deficiencies highlighted some basic features in common.

In short, it was common for researchers in previous HIV cure studies to:
- extract PBMC (peripheral blood mononuclear cells) from the patient and expose them outside the body to agents that could modulate or delete the CCR5.
- target PBMC (leukapheresis) and multipotent CD34+ bone marrow hematopoietic precursor (progenitor) cells.
- use viral vectors such as lentivirus, retrovirus, and adenovirus, or naked RNA transfection, to introduce the desired gene therapy.
- achieve improved acceptance of the cells by reduced-intensity conditioning with a low dose of a chemotherapy drug that works by slowing or halting cell growth.
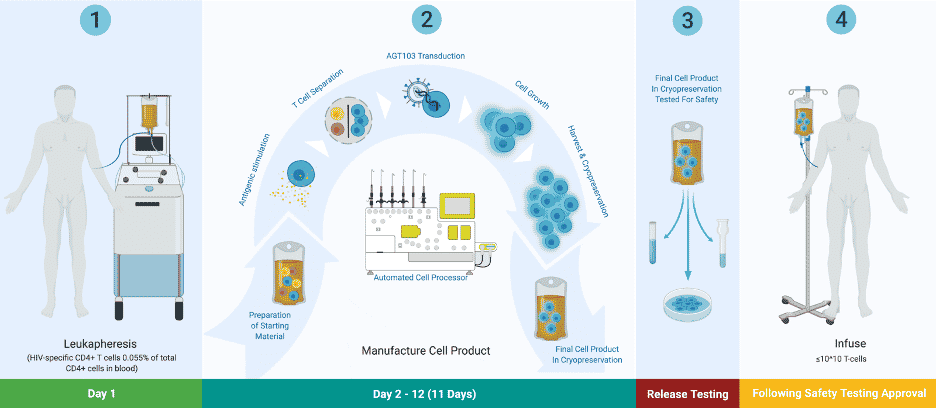
AGT103-T Process
AGT103-T is a cell and gene immunotherapy for HIV disease. It may achieve a functional cure when delivered in a single dose through an infusion. “This product is designed to reinvigorate the host immune response to HIV,” said Pauza, “to create a natural response that’s capable of attacking HIV and resolving it in much the way we do for many other viral diseases.” Gag-specific (HIV-specific) CD4 T cells are a population of T cells that is depleted early in HIV disease. They are an essential component of a healthy immune system and are not recovered despite prolonged therapy.
AGT103-T is designed to reinvigorate and support a potent antiviral immune response by:
- Reconstituting the Gag-specific CD4 T cells through the infusion of a highly enriched, autologous cell product. These group-specific CD4 T cells are essential to most immunological functions. It is the depletion of this subset of T cells that is responsible for the destruction of the immune system in HIV-positive individuals. Replenishment of the supply of these CD4 T cells is essential if the immune system is to function properly.
- Engineering Gag-specific CD4 T cells to resist HIV infection and inhibit HIV release from latently-infected cells. This is a necessary step because it is not enough to replenish the supply of healthy T cells; the new cells must be able to resist infection and keep cells in the HIV reservoirs from releasing latent HIV. Pauza said, “We engineer T cells in this product to resist HIV infection themselves. This makes them more durable once we place them back in the body and also prevents them from releasing HIV if they were part of the viral reservoir. Our hope is that this product will restore the capacity of generating a natural cytotoxic T cell response and also may promote more rapid evolution and production of neutralizing antibodies.”
- Restoring the capacity for generating cytotoxic CD8 T cell neutralizing antibodies. CD8 T cells are a critical subpopulation of T cells. They act as a go-between for adaptive immunity, and cytotoxic T cells are essential for killing HIV-infected cells. Further, the gag-specific CD4 T cells should work with B cells to make neutralizing antibodies.
Successful Manufacturing Process
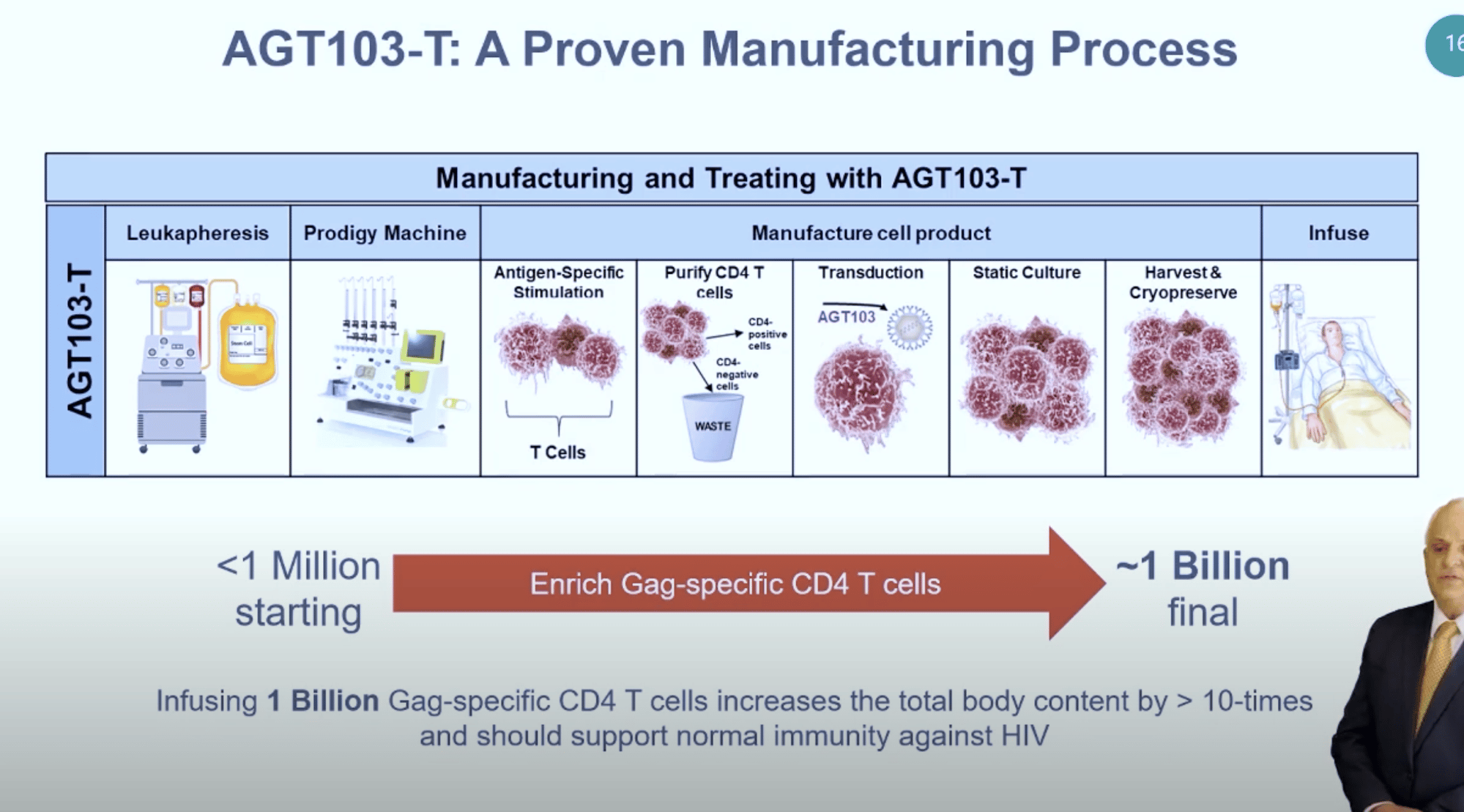
The results from AGT103-T are encouraging. By enriching Gag-specific CD4 T cells (a departure from previous HIV gene therapy studies that were reviewed) AGT achieved approximately 1 billion Gag-specific CD4 T cells from a starting point of 1 million Gag-specific CD4 T cells. It is anticipated that infusing these 1 billion cells will increase the total count in the patient by more than 10 times. This should support normal immunity against HIV, resulting in the desired functional cure.
Next Steps
AGT is pursuing reconstitution of the immune system rather than replacement of genetically engineered, bulk CD4 T cells. The reconstitution method is used to overcome a key immunological defect by infusing an enriched cell population. In the case of HIV, the defect is irreparable loss of HIV-specific CD4 T cells and the failure to recover these cells despite prolonged antiretroviral therapy.
When compared with the SB-728-T (Sangamo Biosciences) study that used zinc finger nuclease anti-CCR5, AGT103-T’s use of the lentivirus vector LV AGT103 returned 2,000 times the dose of Gag-specific CD4 T cells depleted more completely of CCR5. Specifically, where SB-728-T delivered ~ 3.5x105 Gag-specific CD4 T cells, AGT103-T delivered ~ 8.5x108 Gag-specific CD4 T cells. The next step is for AGT to test AGT103-T in a clinical setting.
Take away:
American Gene Technologies (AGT) is determined to restore active HIV immunity to individuals infected with HIV. To that end, they have developed a promising approach. In 2016, AGT met with the FDA to propose a new approach for treatment.In 2019, AGT submitted an Investigational New Drug (IND) application to the FDA, seeking approval to begin a human trial.
In 2020, NIAID repeated AGT’s approach to a functional cure for HIV/AIDS.. Researchers at NIAID were able to replicate AGT’s results. Their findings are documented in a peer-reviewed article co-authored by AGT and published in the May issue of Molecular Therapy. AGT recently began its Phase 1 study. The clinicaltrials.gov identifier number is NCT04561258 and the study ID is AGT-HC168. For information about the ongoing Phase 1 study of AGT103-T, and information on the trial sites, click here.
AGT Founder Jeff Galvin describes what AGT’s trial will accomplish as “ isolating HIV T-cells in somebody's body to make them immune to HIV…. I'm improving the function of those cells, so they're able to clear HIV without becoming infected, just the way your cold T-cells can clear a cold without becoming infected by the cold, which is the key to clearing this pathogen? It’s that simple.”









