The Arc of HIV History


Contributing Author John Vandermosten, Senior Biotechnology Analyst
The Early Days When We Knew Nothing
It is uncertain how the human immunodeficiency virus or HIV initially infected humans although there are educated guesses. Research has suggested that the virus crossed species from chimpanzees to humans in Africa early in the 20th century. Hunters would target chimpanzees or monkeys and consume their meat or become exposed to their blood, and it is speculated that it was through this conduit that the pathogen made its way into humans. Chimps and other primates carry what is called the simian immunodeficiency virus (SIV), which is closely related to HIV and it is hypothesized that through an unfortunate mutation, the virus jumped species into humans. While primates are able to keep the immunodeficiency virus in check as they have had thousands of years to adapt, humans are not so fortunate. The earliest verified infection in humans was found in a man who died in Kinshasa, Democratic Republic of the Congo over 60 years ago. The assessment came from a blood sample collected in 1959 and tested for HIV in the late 1990s.
Kinshasa (Leopoldville) 19591

Through commercial routes and the sex trade, the HIV virus spread from the Congo in the first part of the 20th century throughout the world where it was largely overlooked until the 1980s when there were multiple reports in major US cities of a mysterious illness. The illness was associated with unusual infections such as pneumonia and a certain type of cancer called Kaposi’s sarcoma, which produced highly visible purplish spots on the skin. In July 1981 the New York Times ran an article that identified 41 cases of a new type of cancer that had been diagnosed in homosexual men in New York and California. The initial assessment of the patients’ immunological system found that they had severe defects in their T and B cell lymphocytes, which was thought to allow the infections and cancer to spread.

By the end of 1981 there were over 330 cases reported of the new disease and over a third of the victims had died. This spurred the US Congress to take note, launch hearings and call for AIDS research funding. At this point in time, the underlying illness remained a mystery and it was not until May 1983 when Pasteur Institute scientist Dr. Francoise Barré-Sinoussi2 reported the discovery of a retrovirus. The researcher was measuring retroviral activity associated with the enzyme reverse transcriptase. As she notes in her Nobel Prize biography after repeated efforts to isolate the virus, the team decided to use a lymph node biopsy from which lymphocytes were isolated and cultured.
The first week of sampling did not show any reverse transcriptase activity, but in the second week I detected weak enzymatic activity, which increased significantly a few days later. The reverse transcriptase activity level dropped dramatically however, as the T lymphocytes in the culture were dying. To save the culture, with the hope of preserving the virus, we decided to add lymphocytes from a blood donor to the cell culture. This idea proved successful, and as we had hoped, the virus − which was still present in the cell culture − started to infect the newly added lymphocytes and we were soon again able to detect significant reverse transcriptase activity.3
Dr. Barré-Sinoussi 4

The discovery by Dr. Barré-Sinoussi was confirmed by Dr. Robert Gallo in 1984 when he and his team published four papers in Science where identified the virus as human T-cell lymphotropic virus type III (HTLV-III).5 Dr. Gallo and his team later developed an HIV blood test which was approved by the FDA in March 1985 and allowed health workers to screen the national blood supply and identify infected patients.
CCR5: The Deadbolt on the Front Door
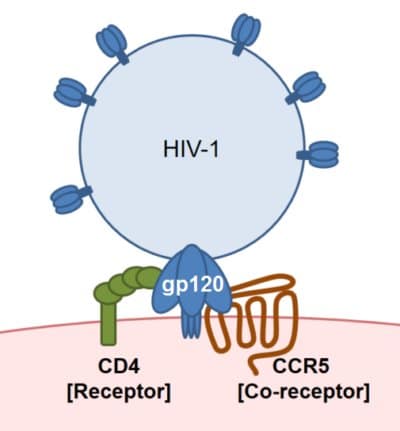
C-C chemokine receptor type 5, also known as CCR5, is a protein on the surface of white blood cells that acts as a receptor. CCR5 plays a role in inflammatory and autoimmune functions directing the flow of chemokines to areas of inflammation or stimulating T cell function and enhancing the adaptive immune response. Despite this mundane existence, the receptor is the entryway for HIV into T cells. CCR5 is the lock that is opened by HIV’s gp120 key which allows the virus to gain a foothold in the human body. The receptor appears on a variety of cells including macrophages, dendritic cells and memory T cells in the immune system. However, HIV specifically targets CD4+ T cells in its efforts to propagate.
CCR5 Receptor Binding to HIV6
Takeover
HIV is a cunning virus that is able to attack and disarm the very immune cells that should kill it. The invader infects CD4+ (T helper) cells and orchestrates a hostile takeover using the cells’ own machinery to propagate and spread further. HIV attaches to a CD4 T cell and fuses with it often via the CCR5 co-receptor, then inserts its genetic material. The reverse transcriptase payload is injected into the CD4+ T cell which converts the accompanying single stranded RNA into double stranded DNA which then travels to the nucleus where it integrates into the host cell chromosome. When cellular machinery synthesizes viral DNA, it sends mRNA out of the nucleus to be translated into viral proteins that will move to the surface of the CD4+ T cell to create a new virion.
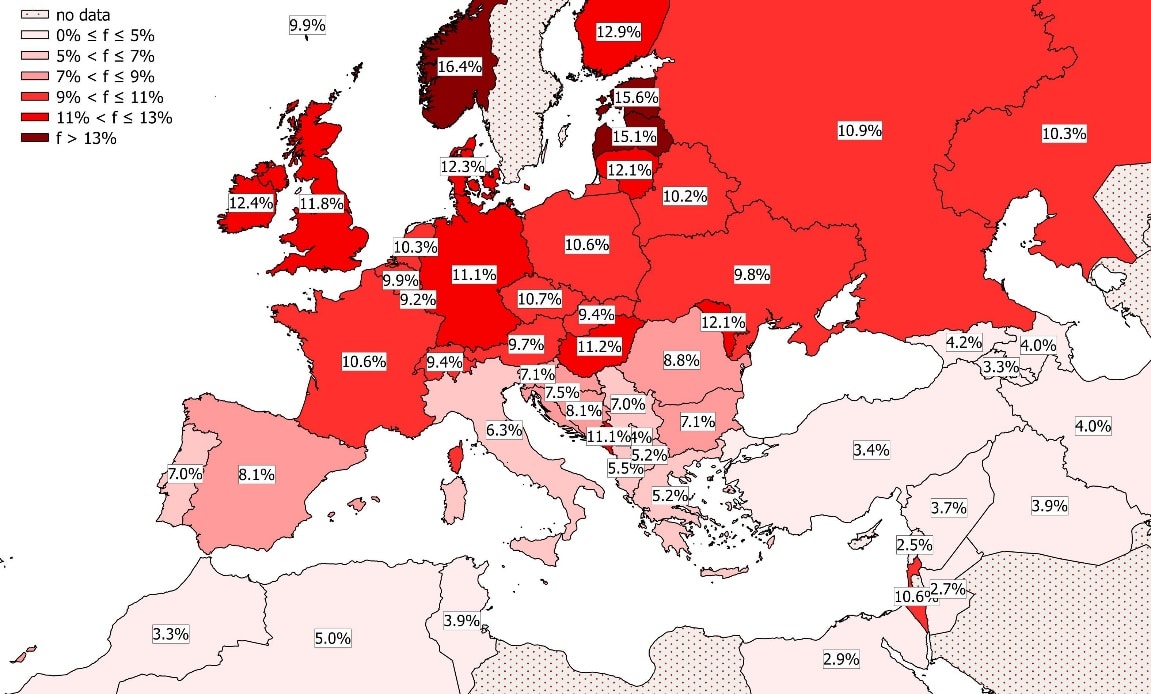
HIV is not the only virus that uses the CCR5 receptor to gain access to a host cell. Looking back centuries and millennia, smallpox and the Plague used this method of entry as did even earlier unidentified viruses that were prevalent up to 5,000 years ago. Because of exposure to these pathogens many thousands of years ago, natural selection has increased the prevalence of the CCR5Δ32 deletion in humans. This mutation prevents the CCR5 receptor from being fully expressed on the CD4+ cell membrane, thereby preventing HIV from entering. The Δ32 mutation occurs in about 10% of those of European descent, either in homozygous or heterozygous form. It is estimated that about 1% of individuals are biallelic and can fully prevent HIV from entering the cell. The rest have heterozygous genes that confer partial protection from HIV in some patients and slow the disease’s advance.
Exhibit I – CCR5-Δ32 Allele Frequency in Europe7
Resistance Isn’t Futile
In 1994, the first person resistant to HIV was discovered. During the early 1990s a researcher named Dr. William Paxton at the Aaron Diamond AIDS Research Center was studying individuals that had been exposed to HIV but had not become infected. Dr. Paxton heard through his network of an individual who had likely been exposed multiples of times to HIV but had not progressed to AIDS and immediately asked him to contribute a blood sample. The scientist began testing the patient’s samples but was surprised at not being able to infect the subject’s CD4+ white blood cells, even when using an extremely high concentration of HIV. The volunteer that provided the immune cells was Steve Crohn, a New York based artist and editor who felt there was something different about himself as those close to him suffered and died from the diseases while he remained healthy. Mr. Crohn had serendipitously inherited two copies of the Δ32 mutation which inactivated the CCR5 receptor thereby locking out the HIV infection and providing a clue to a vulnerability of the virus.
This finding demonstrated that individuals without the CCR5 receptor could block HIV by preventing viral access to target cells, germinating an idea to successfully fight the disease. Understanding the role of CCR5 would eventually lead to the first functional cure of HIV following a bone marrow transplant and spur research and clinical trials using gene therapy to render an HIV patient’s CCR5 receptors defective. We will walk through the next stage of the arc of HIV history introducing the Berlin patient in our next installment and also highlight the antiviral treatments that radically improved survival for the HIV population.
1 Source: History in Orbit, https://historyinorbit.com/photos-of-cities-past-and-present/15
2 Dr. Barré-Sinoussi would go on to win the Nobel Prize in Physiology or Medicine in 2008 for her discovery of HIV which she shared with Luc Montagnier for their discovery of human immunodeficiency virus.
3 Source: https://www.nobelprize.org/prizes/medicine/2008/barre-sinoussi/facts/
4 Source: Nobel Prize website. https://www.nobelprize.org/womenwhochangedscience/stories/francoise-barre-sinoussi
5 We would be remiss if we did not mention the early contention and later resolution of which team, the French or the US, first discovered the virus.
6 Kriner, M., Comparing human and monkey proteins reveals determinants of HIV-1 host cell entry. DECEMBER 18, 2017. Image provided by John Nahabedian.
7 Sollloch, U.V., et al. Frequencies of gene variant CCR5-Δ32 in 87 countries based on next-generation sequencing of 1.3 million individuals sampled from 3 national DKMS donor centers. Human Immunology Volume 78, Issues 11–12, November 2017, Pages 710-717





